RINVOQ vs
HUMIRA® (adalimumab)
SELECT-SWITCH Study in RA
Explore head-to-head data for RINVOQ vs HUMIRA in moderate to severe rheumatoid arthritis patients who have had an inadequate response or intolerance to 1 TNFi.
US-MULT-250253
WARNING: Serious Infections, Mortality, Malignancies, Major Adverse Cardiovascular Events, and Thrombosis
INDICATION
RINVOQ is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) who have had an inadequate response or intolerance to one or more tumor necrosis factor (TNF) blockers.
Limitations of Use: RINVOQ is not recommended for use in combination with other Janus kinase (JAK) inhibitors, biologic disease-modifying antirheumatic drugs (bDMARDs), or with potent immunosuppressants such as azathioprine and cyclosporine.
SELECT-SWITCH Study Design Intro: 12-week, randomized, double blind, head-to-head (H2H), Phase 3b/4 study that evaluated the efficacy and safety of RINVOQ (upadacitinib) + MTX versus HUMIRA (adalimumab) + MTX in 491 adult patients with moderate to severe RA who demonstrated prior failure to one non-adalimumab TNF inhibitor. Patients were randomized to receive RINVOQ 15 mg once daily + MTX or HUMIRA 40 mg every other week + MTX for 12 weeks. The primary endpoint was the proportion of patients who achieved LDA (based on DAS28-CRP ≤3.2) at Week 12.2,3
Please see RINVOQ Important Safety Information, including BOXED WARNING on Serious Infections, Mortality, Malignancies, Major Adverse Cardiovascular Events, and Thrombosis, below.
Click to see HUMIRA® (adalimumab) Indications and Important Safety Information, including BOXED WARNING for Serious Infections and Malignancy. See full HUMIRA Prescribing Information.
SUPERIOR LDA & REMISSION* vs HUMIRA (adalimumab)
in patients with an inadequate response or intolerance to 1 TNFi2
SELECT-SWITCH: A head-to-head study in a TNFi-IR patient population evaluating two products with different benefit/risk profiles
LDA: DAS28-CRP ≤3.2 (NRI-MI, primary endpoint)
Remission: DAS28-CRP <2.6 (NRI-MI, ranked secondary endpoint)
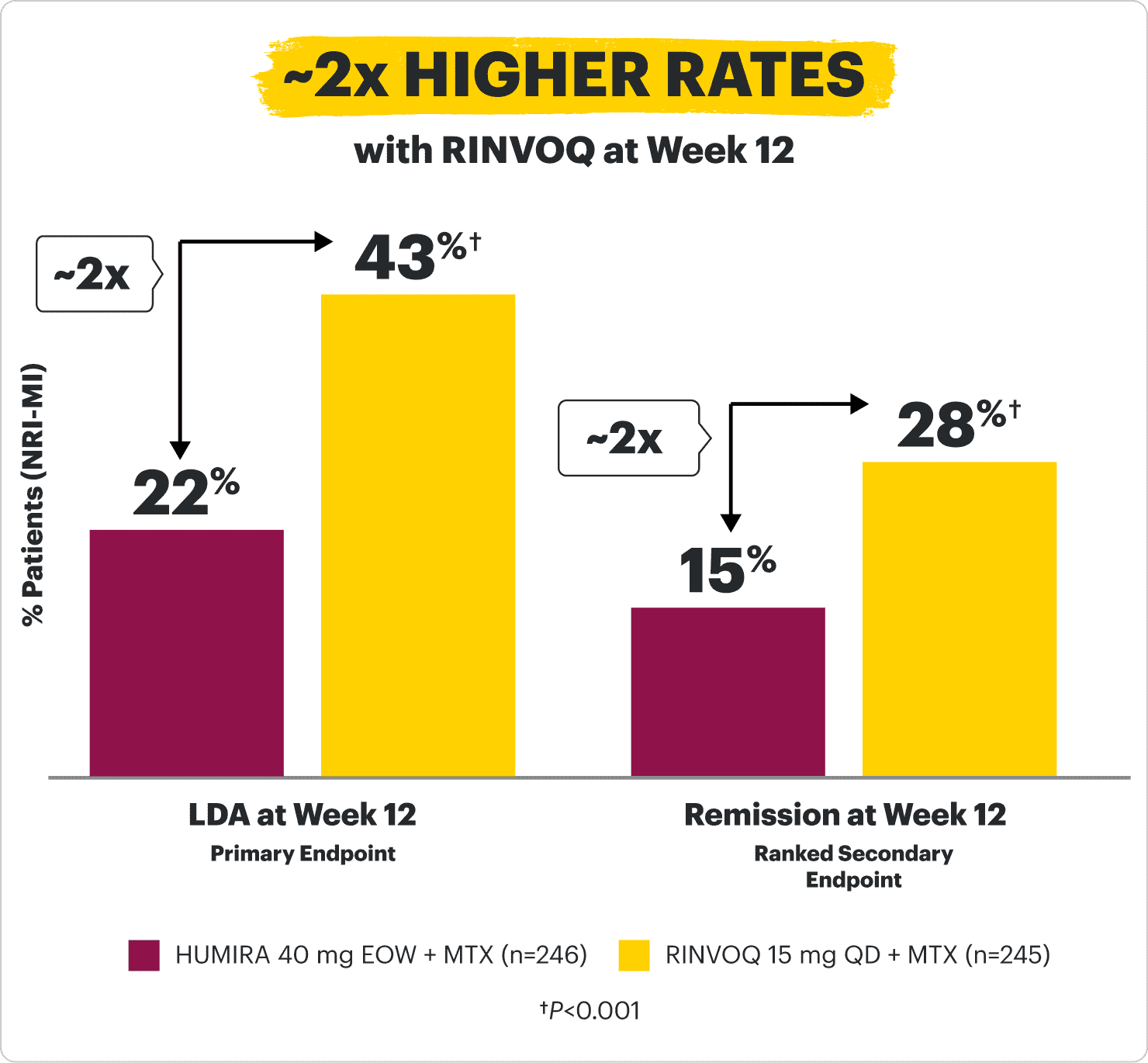
*Clinical remission does not mean drug-free remission or complete absence of disease activity.
RINVOQ is indicated for moderate to severe RA in adult TNFi-IR patients.
RINVOQ has a Boxed Warning on Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis.
HUMIRA is indicated in adults with moderate to severe active RA.
HUMIRA has a Boxed Warning for Serious Infections and Malignancy.
Details about these Boxed Warnings and other risks can be found throughout.
Clinical decisions regarding treatment selection should take into account all relevant information, including full benefit/risk profiles in each product’s PI.

SUPERIOR REMISSION* RATES AT WEEK 12 WITH
RINVOQ vs HUMIRA (adalimumab)
in patients with an inadequate response or intolerance to 1 TNFi2
SELECT-SWITCH: A head-to-head study in a TNFi-IR patient population evaluating two products with different benefit/risk profiles
Remission: DAS28-CRP <2.6 (NRI-MI, ranked secondary endpoint)
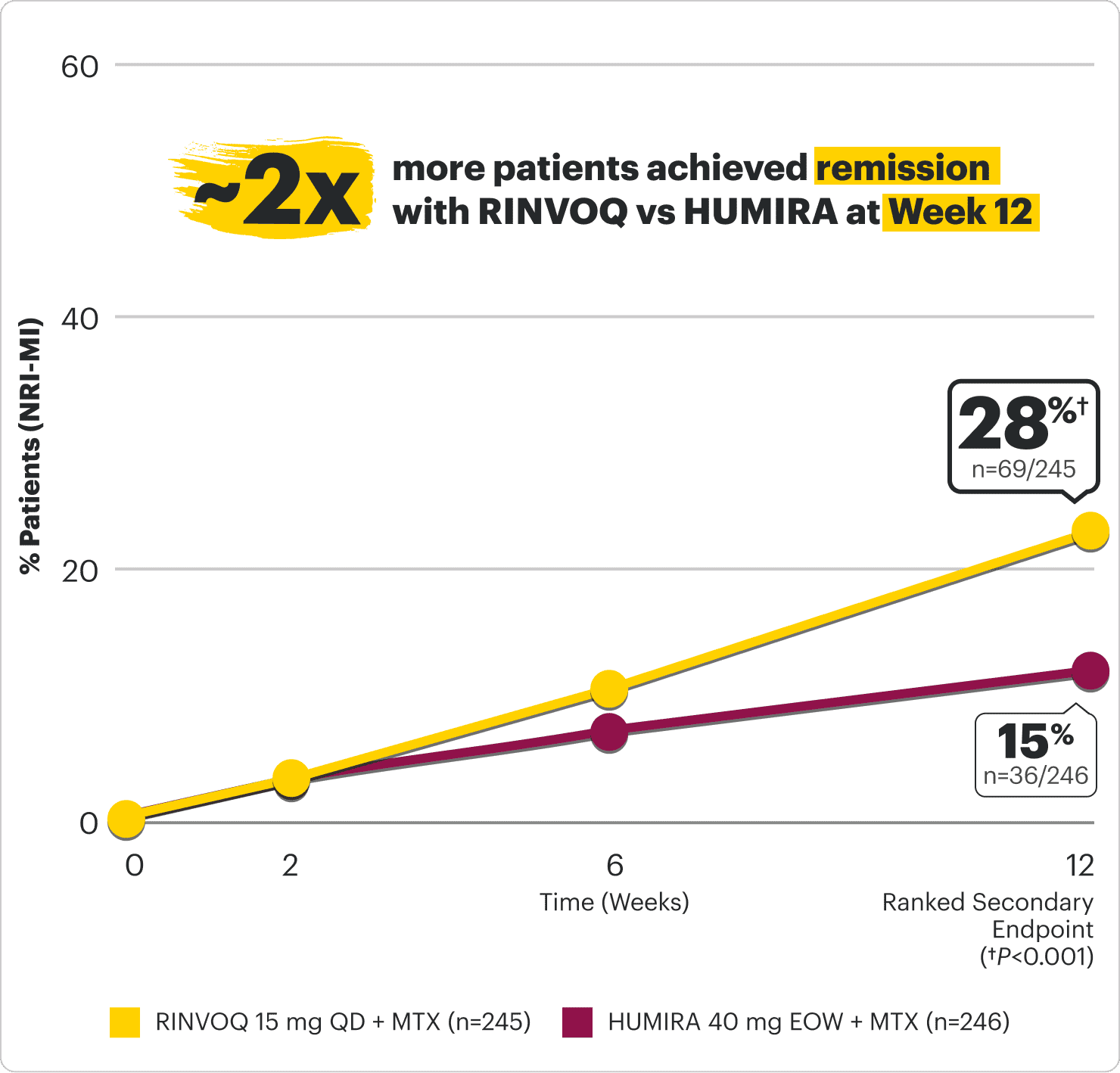
*Clinical remission does not mean drug-free remission or complete absence of disease activity.
RINVOQ is indicated for moderate to severe RA in adult TNFi-IR patients.
RINVOQ has a Boxed Warning on Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis.
HUMIRA is indicated in adults with moderate to severe active RA.
HUMIRA has a Boxed Warning for Serious Infections and Malignancy.
Details about these Boxed Warnings and other risks can be found throughout.
Clinical decisions regarding treatment selection should take into account all relevant information, including full benefit/risk profiles in each product’s PI.
DATA LIMITATIONS: Data not labeled as a ranked secondary endpoint were not controlled for multiplicity; therefore, differences could represent chance findings. No conclusions regarding these comparisons can be made.
SUPERIOR LDA RATES AT WEEK 12 WITH
RINVOQ vs HUMIRA (adalimumab)
in patients with an inadequate response or intolerance to 1 TNFi2
SELECT-SWITCH: A head-to-head study in a TNFi-IR patient population evaluating two products with different benefit/risk profiles
LDA: DAS28-CRP ≤3.2 (NRI-MI, primary endpoint)
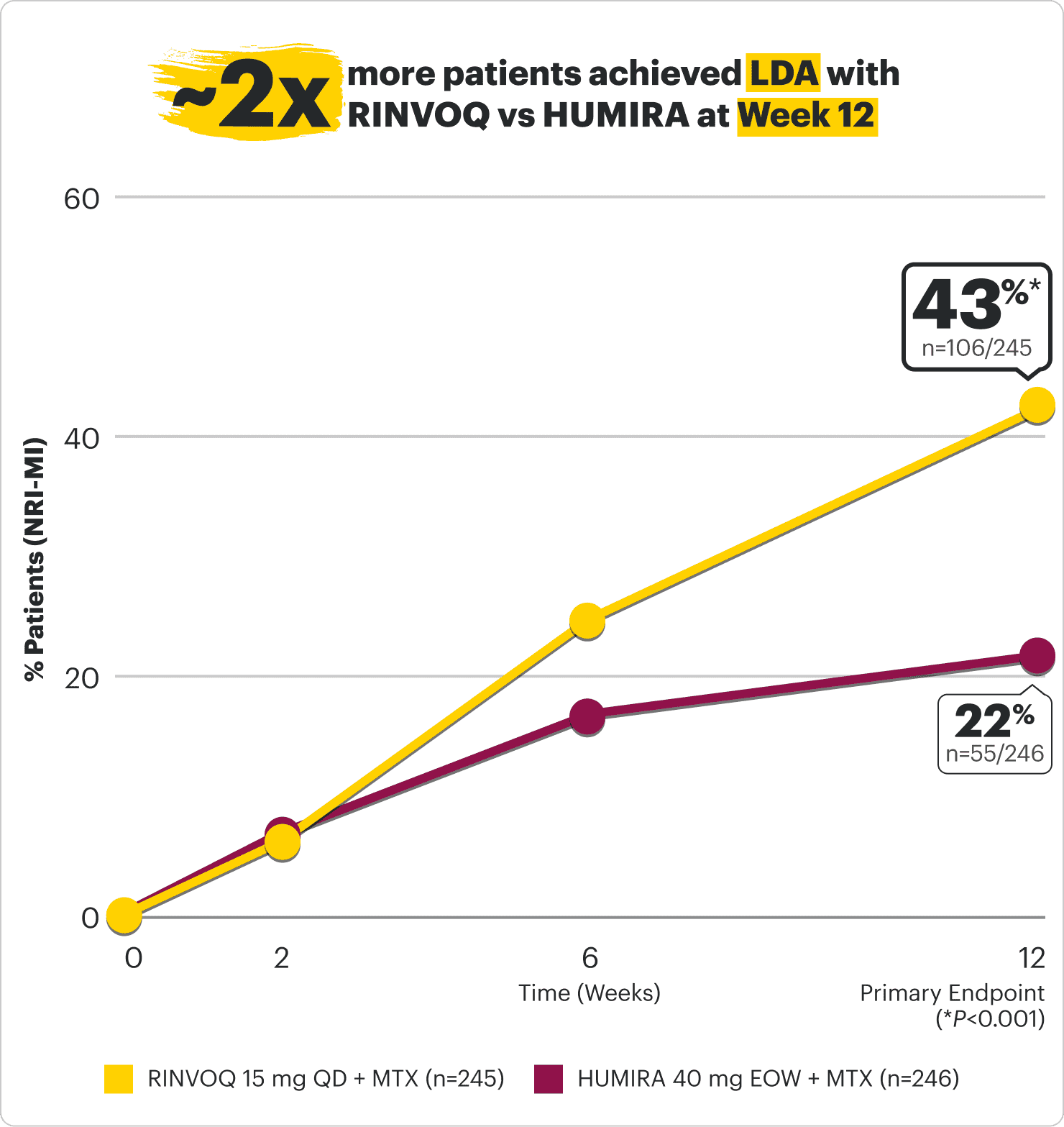
RINVOQ is indicated for moderate to severe RA in adult TNFi-IR patients.
RINVOQ has a Boxed Warning on Serious Infections, Mortality, Malignancy, Major Adverse Cardiovascular Events, and Thrombosis.
HUMIRA is indicated in adults with moderate to severe active RA.
HUMIRA has a Boxed Warning for Serious Infections and Malignancy.
Details about these Boxed Warnings and other risks can be found throughout.
Clinical decisions regarding treatment selection should take into account all relevant information, including full benefit/risk profiles in each product’s PI.
DATA LIMITATIONS: Data not labeled as a ranked secondary endpoint were not controlled for multiplicity; therefore, differences could represent chance findings. No conclusions regarding these comparisons can be made.

Serious Infections: RINVOQ-treated patients are at increased risk of serious bacterial (including tuberculosis [TB]), fungal, viral, and opportunistic infections leading to hospitalization or death. Most patients who developed these infections were taking concomitant immunosuppressants, such as methotrexate or corticosteroids.
Mortality: A higher rate of all-cause mortality, including sudden cardiovascular (CV) death, was observed with a Janus kinase inhibitor (JAKi) in a study comparing another JAKi with tumor necrosis factor (TNF) blockers in rheumatoid arthritis (RA) patients ≥50 years with ≥1 CV risk factor.
Malignancies: Malignancies have occurred in RINVOQ-treated patients. A higher rate of lymphomas and lung cancer (in current or past smokers) was observed with another JAKi when compared with TNF blockers in RA patients.
Major Adverse Cardiovascular Events: A higher rate of CV death, myocardial infarction, and stroke was observed with a JAKi in a study comparing another JAKi with TNF blockers in RA patients ≥50 years with ≥1 CV risk factor. History of smoking increases risk.
Thromboses: Deep venous thrombosis, pulmonary embolism, and arterial thrombosis have occurred in patients treated for inflammatory conditions with JAK inhibitors, including RINVOQ. A higher rate of thrombosis was observed with another JAKi when compared with TNF blockers in RA patients.
Hypersensitivity: RINVOQ is contraindicated in patients with hypersensitivity to RINVOQ or its excipients.
Other Serious Adverse Reactions: Hypersensitivity Reactions, Gastrointestinal Perforations, Laboratory Abnormalities, and Embryo-Fetal Toxicity.
<<Swipe table to see more
| TEAE of Special Interest* |
SELECT-SWITCH AEs2 | Phase 3 Program AEs4 | |||
| HUMIRA 40 mg EOW + MTX† Week 12 n=245 n (%) |
RINVOQ 15 mg QD + MT† Week 12 n=245 n (%) |
Any RINVOQ 15 mg QD† ~9.5 yrs max exposure‡ ~4.2 yrs median n=3209 PYs=12909.0 E/100 PYs unless otherwise stated |
|||
| INFECTIONS | |||||
| Serious infections | 2 (0.8) | 2 (0.8) | 3.5 | ||
| Active Tuberculosis (TB) | 0 | 0 | <0.1 | ||
| Opportunistic Infection (excluding TB, HZ, & oral candidiasis) | 1 (0.4) | 0 | 0.2 | ||
| Herpes Zoster (HZ) | 4 (1.6) | 1 (0.4) | 3.0 | ||
| MALIGNANCY | |||||
| Malignancy (excluding NMSC) | 1 (0.4) | 1 (0.4) | 0.7c | ||
| Lymphoma | 0 | 0 | <0.1c | ||
| NMSC | 0 | 0 | 0.4c | ||
| CARDIOVASCULAR EVENTS | |||||
| Adjudicated VTEa | 0 | 0 | 0.4c | ||
| Adjudicated MACEb | 0 | 0 | 0.4c | ||
| GASTROENTEROLOGICAL EVENTS | |||||
| Adjudicated GI Perforations | 0 | 0 | <0.1 | ||
| LAB ABNORMALITIES | |||||
| The percentages of patients with lab-related AESIs are listed here for HUMIRA and RINVOQ, respectively: anemia (0.4, 1.6), neutropenia (1.2, 0.4), lymphopenia (0.4, 0.8), hepatic disorder (1.6, 2.9) | |||||
An increased risk of death, malignancy (excluding NMSC), MACE, and VTE have been observed in the XELJANZ® (tofacitinib) ORAL Surveillance study, a safety outcomes trial of a different JAK inhibitor plus MTX in rheumatoid arthritis patients >50 years of age with an additional CV risk factor compared to TNFi plus MTX.
The FDA has determined that all JAKis, including topicals, indicated for the treatment of inflammatory conditions, may have similar risks because of shared mechanisms of action. Therefore, each JAKi product's USPI contains a related boxed warning for these risks and thus should be considered in your treatment choices for your patients with moderate to severe RA.
SELECT-SWITCH was not powered or designed to evaluate safety outcomes between RINVOQ and HUMIRA. Certain adverse events may require longer observation periods and longer-term patient exposure to ascertain risk. Thus, this presentation does not mean that the risks detected in the ORAL Surveillance study would not be seen in RINVOQ.
Safety rates observed in clinical trials may not predict the rates observed in clinical practice. No comparisons can be made between the RINVOQ program Phase 3 AEs and SELECT-SWITCH AEs.
RINVOQ requires lab monitoring for neutropenia, lymphopenia, anemia, increased lipids, and AST/ALT elevation whereas HUMIRA does not.
RINVOQ is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) who have had an inadequate response or intolerance to one or more tumor necrosis factor (TNF) blockers.
Limitations of Use: RINVOQ is not recommended for use in combination with other Janus kinase (JAK) inhibitors, biologic disease-modifying antirheumatic drugs (bDMARDs), or with potent immunosuppressants such as azathioprine and cyclosporine.
REFERENCES:
RINVOQ vs
HUMIRA® (adalimumab)
SELECT-SWITCH Study in RA
Explore head-to-head data for RINVOQ vs HUMIRA in moderate to severe rheumatoid arthritis patients who have had an inadequate response or intolerance to 1 TNFi.